Hemorrhoidal staplers have become an important surgical device in modern proctology, especially for the treatment of prolapsed and internal hemorrhoids. These devices support minimally invasive procedures that help surgeons achieve precise results while reducing patient discomfort and recovery time.
As healthcare providers continue to prioritize safety, efficiency, and consistent surgical outcomes, the demand for high-quality hemorrhoidal staplers has increased significantly. Choosing a reliable manufacturer is essential to ensure product performance, regulatory compliance, and dependable supply. Universal Sutures is committed to delivering advanced hemorrhoidal staplers designed to meet the evolving needs of hospitals, surgeons, and medical distributors worldwide.
What is a Hemorrhoidal Stapler?
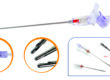
A hemorrhoidal stapler is a specialized surgical instrument used in stapled hemorrhoidopexy procedures. The device removes excess hemorrhoidal tissue and repositions the remaining tissue to restore its normal anatomical structure. This method is widely preferred because it reduces surgical trauma and supports faster patient recovery compared to conventional hemorrhoid surgery.
Key Benefits of Using Hemorrhoidal Staplers
Reduced postoperative pain
Shorter hospital stay and faster recovery
Minimal tissue damage
Lower risk of complications
Improved patient comfort and satisfaction
These benefits make hemorrhoidal staplers a valuable tool in modern colorectal surgery.
Trusted Hemorrhoidal Stapler Manufacturer
Universal Sutures is a recognized manufacturer of high-quality surgical devices designed to deliver reliable performance in clinical settings. Our manufacturing facilities are equipped with modern technology and operated by skilled professionals who follow strict quality and safety standards.
We focus on producing medical devices that meet global healthcare requirements while ensuring consistent product quality and patient safety. Our experience in surgical device manufacturing enables us to provide dependable solutions for hospitals, surgical centers, and healthcare distributors.
Our Manufacturing Capabilities Include
Advanced production infrastructure
Use of medical-grade materials
Precision engineering and assembly
Strict quality control procedures
Compliance with international standards
These capabilities allow us to maintain consistent product reliability across every batch we manufacture.
Advanced Manufacturing and Quality Assurance
The production of hemorrhoidal staplers requires a controlled manufacturing environment and detailed quality verification at every stage. At Universal Sutures, we follow a structured process designed to ensure device safety, durability, and performance.
The process begins with product design and engineering, where the device is developed to provide ergonomic handling and accurate stapling performance. High-quality materials are then carefully selected to ensure strength, compatibility, and long-term reliability.
Each device is assembled using precision equipment and trained technicians to maintain consistent functionality. After assembly, the products undergo multiple inspections and functional tests to verify mechanical reliability, staple formation accuracy, and sterility.
Finally, the devices are sterilized and securely packaged to protect product integrity during storage and transportation.
Key Features of Our Hemorrhoidal Staplers
Universal Sutures hemorrhoidal staplers are designed to support surgeons with dependable performance and ease of use in clinical environments.
Product Features
Smooth and reliable firing mechanism
Ergonomic and surgeon-friendly design
Consistent and secure staple formation
High tissue compatibility
Reduced risk of tissue trauma
Sterile, ready-to-use packaging
Long shelf life and reliable performance
These features help healthcare professionals perform procedures efficiently while maintaining patient safety.
Quality Standards and Regulatory Compliance
Maintaining strict quality standards is essential in medical device manufacturing. Universal Sutures follows internationally recognized quality management systems to ensure product safety and regulatory compliance.
Our manufacturing processes align with global medical device requirements and industry best practices. This commitment helps healthcare providers trust our products for safe and effective surgical use.
Compliance Standards
- ISO 13485 Quality Management System
- CE Marking requirements
- Good Manufacturing Practices (GMP)
- International medical device safety standards
- By adhering to these standards, we ensure consistent product quality and reliability across all markets.
Applications of Hemorrhoidal Staplers
Hemorrhoidal staplers are widely used in hospitals, surgical centers, and specialty clinics for the treatment of various anorectal conditions. These devices support efficient surgical procedures and improved patient outcomes.
Common Uses
- Prolapsed hemorrhoids
- Internal hemorrhoids
- Stapled hemorrhoidopexy procedures
- Rectal mucosal prolapse treatment
- Minimally invasive colorectal surgeries
Their versatility makes them an essential device in modern surgical practice.
Conclusión
Hemorrhoidal stapler manufacturing requires precision engineering, strict quality control, and a strong understanding of surgical requirements. Universal Sutures combines advanced technology, skilled expertise, and reliable production systems to deliver high-quality hemorrhoidal staplers for healthcare providers worldwide.
With a focus on safety, performance, and consistent supply, Universal Sutures remains a trusted partner for hospitals and medical distributors seeking dependable surgical solutions.